INTERIORDECOR.BIZ.ID – Light is a fundamental aspect of our universe, playing a crucial role not only in how we perceive the world but also in the very fabric of chemical reactions. From initiating life-sustaining processes on Earth to driving complex chemical transformations in laboratories, light‘s influence is profound and far-reaching. Understanding light’s definition in chemistry unlocks a deeper appreciation for numerous scientific phenomena and technological applications.
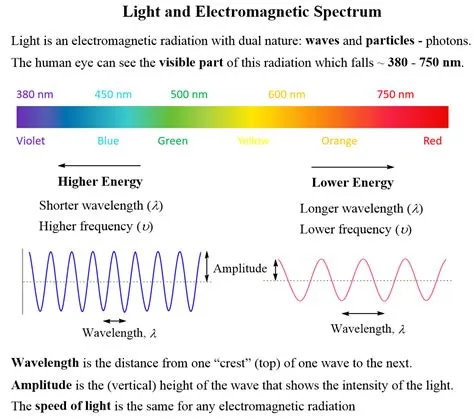
In its most basic definition within chemistry and physics, light is a form of electromagnetic radiation. This radiation travels in waves and also exhibits particle-like properties, behaving as discrete packets of energy called photons. The electromagnetic spectrum encompasses a vast range of wavelengths, and visible light represents only a small portion that our eyes can detect.
The Electromagnetic Spectrum and Light
The electromagnetic spectrum organizes all types of electromagnetic radiation by their wavelength and frequency. This spectrum includes radio waves, microwaves, infrared radiation, visible light, ultraviolet (UV) radiation, X-rays, and gamma rays. Each type of radiation has a unique energy level associated with its photons, determined by its frequency.
Visible light, the part of the spectrum we perceive as color, has wavelengths ranging from approximately 400 nanometers (violet) to 700 nanometers (red). Different wavelengths within this range correspond to different colors, and our eyes interpret these variations. This ability to differentiate colors is a direct consequence of how light interacts with matter.
Interaction of Light with Matter
When light encounters matter, several interactions can occur, each with significant chemical implications. These interactions include absorption, reflection, transmission, and scattering. The specific outcome depends on the wavelength of the light and the properties of the material it strikes.
Absorption is particularly critical in chemistry, as it describes the process where a substance takes in light energy. This absorbed energy can excite electrons within atoms or molecules, leading to various chemical changes. For example, the green color of plants is due to chlorophyll absorbing red and blue light while reflecting green light.
Photons and Chemical Energy
Photons, the quantum packets of light energy, are central to understanding light’s role in chemistry. The energy of a single photon is directly proportional to the frequency of the light, as described by Planck’s equation (E = hf), where E is energy, h is Planck’s constant, and f is frequency.
When a molecule absorbs a photon, its electrons can jump to higher energy levels. This process is known as electronic excitation. If the absorbed energy is sufficient, it can even lead to the breaking of chemical bonds, initiating a chemical reaction. This is the fundamental principle behind photochemistry.
Photochemistry: Chemistry Driven by Light
Photochemistry is a branch of chemistry that studies chemical reactions initiated by the absorption of light. These reactions are distinct from thermal reactions, which are driven by heat energy. Light provides a highly specific way to deliver energy to molecules, often enabling reactions that are difficult or impossible to achieve through heating alone.
A prime example of photochemistry is photosynthesis, the process by which plants use sunlight to convert carbon dioxide and water into glucose and oxygen. Chlorophyll molecules capture light energy, which then drives the complex series of reactions that sustain plant life and, consequently, much of life on Earth.
Spectroscopy: Analyzing Light’s Interaction
Spectroscopy is a powerful analytical technique in chemistry that utilizes the interaction of light with matter to identify and quantify substances. By measuring how much light a sample absorbs or emits at different wavelengths, scientists can deduce its chemical composition and structure.
Techniques like UV-Vis spectroscopy, infrared (IR) spectroscopy, and atomic emission spectroscopy are indispensable tools in research and industry. They allow chemists to study molecular structures, monitor reaction progress, and ensure product purity by analyzing the unique spectral fingerprints produced by light interactions.
Applications of Light in Chemistry
The applications of understanding light in chemistry are vast and continue to expand. From the development of solar cells that harness light energy to power our world to the design of new pharmaceuticals that utilize light-activated drug delivery systems, light is a key enabler of innovation.
Photocatalysis, where light is used to accelerate chemical reactions with the help of a catalyst, is another growing field. This has significant implications for environmental cleanup, such as breaking down pollutants in water and air. The precise control offered by light makes these processes more efficient and sustainable.
Conclusion
In essence, light in chemistry is understood as electromagnetic radiation, primarily its energetic photons, that can interact with matter to induce chemical changes. Whether it’s driving photosynthesis, enabling analytical techniques, or powering new technologies, light is an indispensable component of chemical science. Its ability to deliver specific amounts of energy makes it a unique and powerful tool for manipulating matter at the molecular level.
The continuous exploration of light’s properties and interactions promises further breakthroughs in our understanding of the chemical world. As we delve deeper into quantum mechanics and molecular behavior, the role of light will undoubtedly become even more central to scientific discovery and technological advancement.
Frequently Asked Questions (FAQ)
- What is light in the context of chemistry?
- In chemistry, light is defined as a form of electromagnetic radiation that exists as waves and discrete energy packets called photons. It carries energy that can interact with atoms and molecules.
- How does light affect chemical reactions?
- Light can initiate or influence chemical reactions by providing the energy needed to excite electrons in molecules. This absorbed energy can lead to bond breaking or forming, a field known as photochemistry.
- What is the difference between visible light and other electromagnetic radiation?
- Visible light is the portion of the electromagnetic spectrum that human eyes can detect, with wavelengths roughly between 400 and 700 nanometers. Other forms like UV or infrared radiation have different wavelengths and energy levels, leading to different types of interactions.
- What is spectroscopy used for in chemistry?
- Spectroscopy uses the interaction of light with matter to identify, quantify, and study the structure of substances. By analyzing how light is absorbed or emitted, chemists can gain detailed information about a sample.
- Can you give an example of a chemical process driven by light?
- Photosynthesis is a prime example, where plants use sunlight to convert carbon dioxide and water into glucose and oxygen. Photocatalysis, used for pollution control, is another application where light drives chemical reactions.
Written by: Emma Johnson